 Is the process spontaneous at 10.00 C Is it spontaneous at +10.00 C Solution We can assess the spontaneity of the process by calculating the entropy change of the universe. $$\Delta S_$$Ĭonclusion: Although the change in entropy from state 1 to state 2 is the same, less work is done by the irreversible expansion than the reversible expansion due to the entropy generated within the system during the irreversible process. The entropy change for the process H 2 O ( s) H 2 O ( l) is 22.1 J/K and requires that the surroundings transfer 6.00 kJ of heat to the system. But this is only one example of a spontaneous. 
So, from the first law of thermodynamics, the change in internal energy of the combined system is zero. The two containers are presumably insulated, so no heat Q is exchanged with the surroundings. So the change in entropy between two equilibrium states 1-2 is (8.1c), the change in entropy does involve some quantity of heat transferred as well as the temperature. The total volume of the two rigid containers does not change, so the combined system does no work W on the surroundings. A differential change in entropy is defined for a reversible transfer of heat as follows 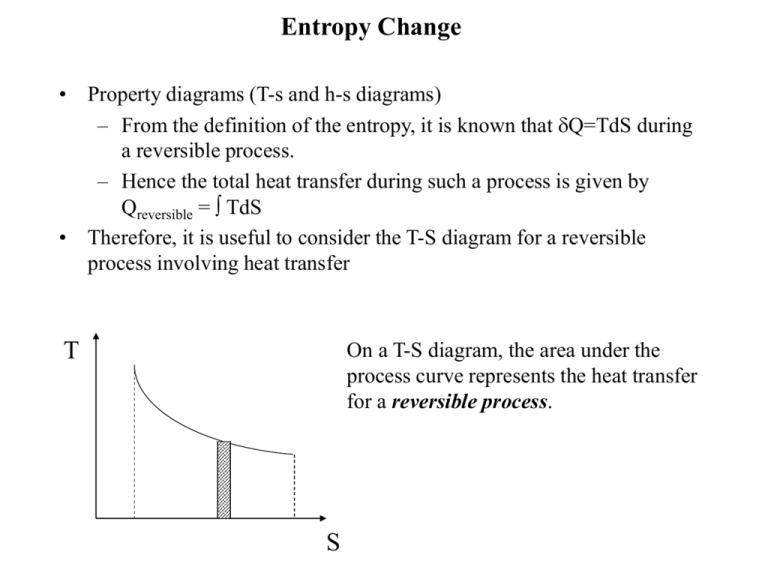
The thermodynamic definition for.entropy change is only valid for 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
