 
Decreases in fluid load are commonly referred to as dehydration.Įdema manifests as swelling in the soft tissues of the limbs and face with a subsequent increase in size and tightness of the skin. Edema occurs most commonly in soft tissues of the extremities however, it is possible to occur in any tissue. Fluid overload is clinically known as edema. Fluid balance abnormalities are either an overload of fluid or a decrease in effective fluid. The combined “push” of hydrostatic forces and the “pull” of osmotic forces create a net movement of fluid. This is mathematically explained using the Starling equation:Ī variety of pathological conditions induce abnormalities in fluid balance. Hydrostatic pressure is the “push” factor on fluid movement where increased pressures force fluid out of a space. In addition to the osmotic pull of fluids, fluid movement within the body relies on created and maintained hydrostatic pressures. This is best utilized in the movement of fluid from plasma in the extracellular blood space into the interstitial spaces of tissue across the capillary membrane. These processes create a cellular environment to osmotically “pull” water into fluid compartments. Solute gradients can be physiologically created by membrane pumping proteins, which expend energy in the form of ATP to move components from areas of low concentration into higher concentrations against their diffusion gradient. If a membrane is non-permeable to an ion, this creates a gradient of relatively higher concentration osmolarity. Ionic components will diffuse through fluids selectively depending on the presence of permeable membranes. All body fluids should have an ionic net electrical charge close to zero, indicating a balance of cations and anions. Fluid will move towards hyperosmotic compartments and away from hypoosmotic compartments. Another important osmotically active component to consider is glucose. In addition to ionic components, osmolarity is partially composed of proteins such as albumin in the serum. Blood plasma osmotic gradients are maintained through the absorption of solutes from the gastrointestinal tract or secretion into the gastrointestinal tract or urine. However, rapid changes in fluid volume without changes in ionic components cause dilation or concentration of those components. Cellular osmotic concentration gradients are maintained largely through the active pumping of transmembrane ionic transport proteins. Less than this is hypoosmotic, and greater is hyperosmotic. Physiologic blood plasma osmolarity is approximately 286 mOsmoles/L. Osmolarity is defined as the number of particles per liter of fluid. įluid moves throughout cellular environments in the body by passively crossing semipermeable membranes. Intracellular fluids tend to be inversed with high levels of phosphate, magnesium, potassium, and proteins but lower sodium, chloride, and bicarbonate. Interstitial fluids physiologically tend to have a low concentration of proteins. Extracellular spaces contain high concentrations of sodium, chloride, bicarbonate, and proteins but are relatively lower in potassium, magnesium, and phosphate. Extracellular fluid and interstitial fluid are similar in composition. 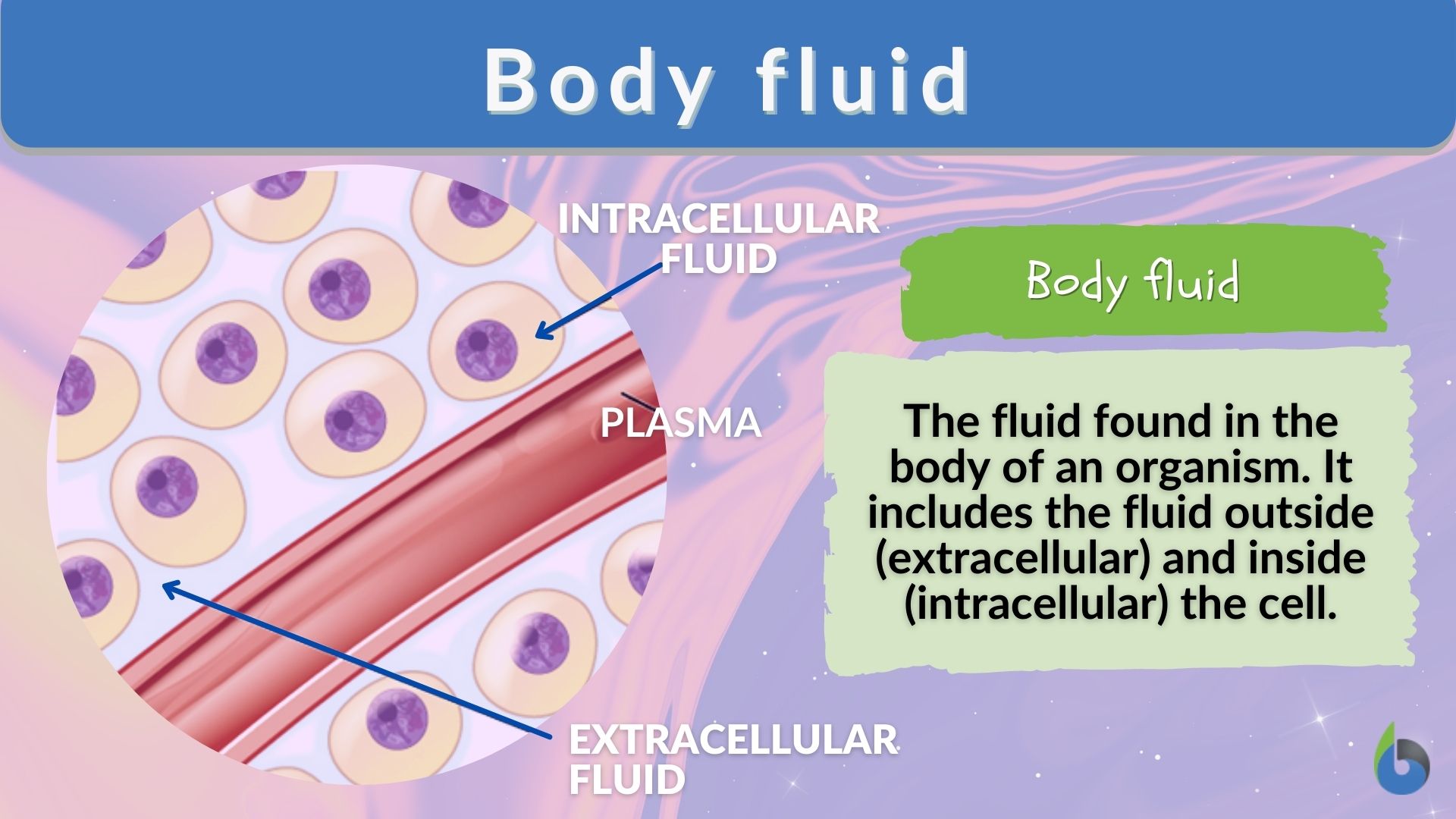
The exact chemical composition of body fluid is highly variable. This is dependent on which portion of the body, as well as which organ of the body, contains the fluid. Additional fluid spaces are possible in pathological scenarios and are categorized as a transudate or exudate based on location and etiology. 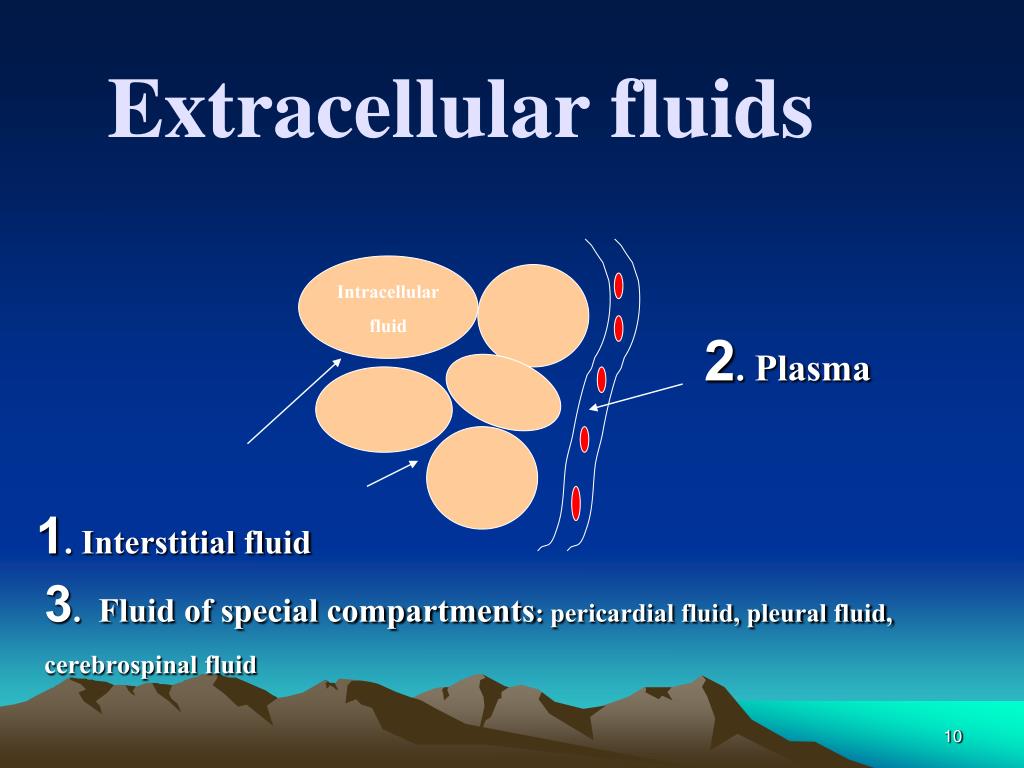
The extracellular fluid comprises approximately 20% of total body weight and further subcategorizes as plasma at approximately 5% of body weight and interstitial space, which is approximately 12% of body weight. As such, it is important to maintain an appropriate osmolality. This space is where many chemical reactions occur. In general, intracellular fluids are stable and do not readily adjust to rapid changes. It is the total space within cells primarily defined as the cytoplasm of cells. Intracellular fluid is approximately 40% of the total body weight. 
The distribution of fluid throughout the body can be broken down into two general categories: intracellular fluid and extracellular fluid. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
